Fall 2019 (Volume 29, Number 3)
Survey Results:
Methotrexate Prescribing Patterns in Canada
By Dr. Shirley Lake, on behalf of the CRA Choosing Wisely sub-committee
Download PDF
This issue’s Joint count survey focused on methotrexate (MTX) prescribing and monitoring patterns in Canada. The survey results show that there is wide variability in how rheumatologists across Canada use MTX in their patients. A total of 126 responses were received from CRA members out of a possible 548, equating to a 23% response rate. Both community and academic rheumatologists responded, representing 27% and 53% of respondents, respectively. Another 20% of respondents said they worked in both settings.
When asked about what tests are ordered when a patient is started on MTX, it seems there are some tests that all rheumatologists order (CBC, creatinine, and ALT), and some that they don’t order (INR), but a great variability in the rest. Recommendation 10 from the CRA guidelines on MTX may provide guidance in this case.1 They state that “A complete blood count (CBC) (level of evidence II), liver (I) and renal biochemistry (II), and a chest radiograph (II) should be ordered prior to initiating MTX therapy. Screening for hepatitis B and C should be considered (III), and HIV testing is recommended in high-risk patients (IV).”1
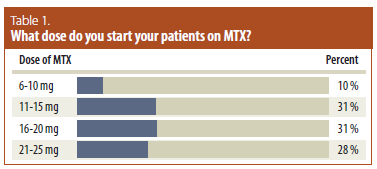
With respect to the starting dose of MTX, there is much variability here as well. Per recommendation 11 of the CRA guidelines, the starting dose should be individualized to the patient based on clinical response and tolerability.1
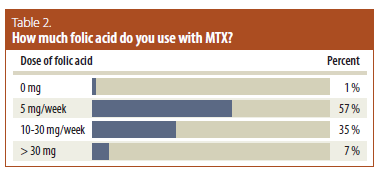
Regarding the dose of folic acid prescribed with MTX, based on the survey results, more than 55% of respondents prescribe 5 mg per week, and more than one third prescribe 10-30 mg per week; another 7% prescribe > 30 mg per week. While the CRA rheumatoid arthritis (RA) management guidelines allow for variation in MTX dosing and routes, they do not provide evidence-based recommendations on folic acid. However, these survey results are consistent with a multinational evidence-based recommendation that states that “Prescription of at least 5 mg folic acid per week with MTX therapy is strongly recommended.” 2
Regarding monitoring for toxicities, the multinational recommendation says that “When starting methotrexate or increasing the dose, ALT (with or without AST), creatinine and CBC should be performed every 1–1.5 months until a stable dose is reached and every 1–3 months thereafter; clinical assessment for side effects and risk factors should be performed at each visit.”2 The majority of survey respondents followed this guideline to check monthly at first, and then every three months. With that said, only three tests need to be monitored (ALT, creatinine, CBC), but many respondents answered that they also monitor serum albumin.
Overall, there seems to be wide variability in how rheumatologists across Canada manage MTX. Physicians who prescribe MTX should be experts in the underlying conditions and in individualizing the dosing and monitoring of MTX; rheumatologists could certainly lead in this area. While the CRA guidelines will be updated, they are still a standard to abide by. The guidelines are available at rheum.ca/resources/publications/canadian-recommendations-for-management-of-ra/.
Ultimately, updated guidelines from the CRA regarding MTX monitoring would be very helpful to guide care–particularly if non-rheumatologists are also doing some of the monitoring of patients in some parts of the country.
For any questions or feedback regarding this survey, contact Sue Ranta at sranta@rheum.ca.
References:
1. Bykerk VP, Akhavan P, Hazlewood GS, et al. Canadian Rheumatology Association recommendations for pharmacological management of rheumatoid arthritis with traditional and biologic disease-modifying antirheumatic drugs. J Rheumatol 2012;39: 1559-82.
2. Visser K, Katchamart W, Loza E, et al Multinational evidence-based recommendations for the use
of methotrexate in rheumatic disorders with a focus on rheumatoid arthritis: integrating systematic
literature research and expert opinion of a broad international panel of rheumatologists in the 3E
Initiative. Annals of the Rheumatic Diseases 2009;68: 1086-93.
|




